|
But for larger nuclei combinations, no energy is released. 
Therefore, the binding of nuclei will release extra energy due to the high attraction between two nuclei. When it comes to tiny nuclei, there are a less number of protons present hence, less repulsion occurs. Since protons are positively charged and repel each other, there should be a strong attractive force to hold them together. The nuclear binding energy is the energy required to hold protons and neutrons together inside the nucleus. The energy released from the sun is a result of nuclear fusion reactions taking place inside the sun. Nuclear fusion reactions are most commonly found in the sun. The difference of mass arises due to the difference in nuclear binding energies. This missing mass is converted into energy. When calculated, there is a mass difference between reactants and products. Sometimes, the nuclei combination will result in more than one large atom. This happens under high temperature and pressure conditions. Nuclear fusion is the combination of two smaller atoms to create a large atom, releasing energy. This energy comes from the nucleus, where the strong nuclear forces between sub-atomic particles are converted into energy. Here, a very high amount of energy is released. If the speed of the bombardment is enough, the two lobes can get separated completely, forming two fragments because the nuclear forces are now not enough to hold the lobes together. This lobe formation causes the sub-atomic particles to separate from each other. This causes the formation of a shape with two lobes. When the nucleus captures the accelerated neutron, the spherical shape of the nucleus is deformed. The nucleus of an atom gets a spherical shape due to the presence of nuclear forces between sub-atomic particles (neutrons and protons). The nuclear fission occurs through a special mechanism called binary fission. This is called “nuclear chain reaction.” Mechanism – Binary Fission The fission produces more neutrons that can induce other isotopes to undergo nuclear fission. 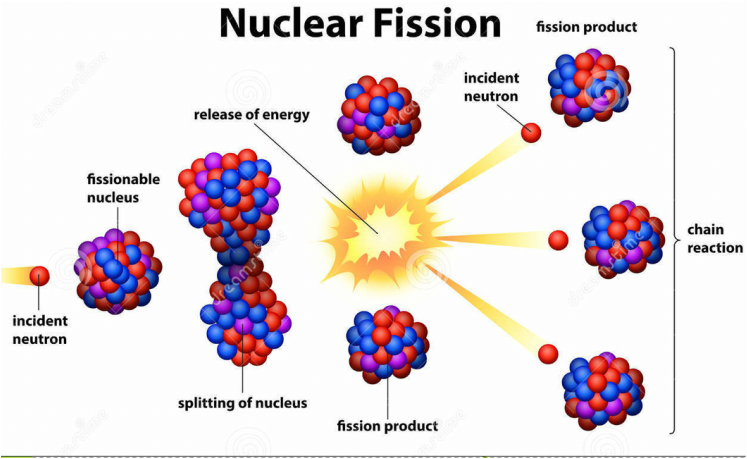
The new nucleus is more unstable thus, it undergoes fission reaction. First, the neutron combines with the nucleus of the isotope. These accelerated neutrons cause the isotope to undergo fission. This is a nonspontaneous reaction where a large, unstable isotope is bombarded with high-speed neutrons. This reaction may occur in two ways as below. A nuclear fission reaction can release a high amount of energy. Often, the products of nuclear fission include neutrons and gamma rays. These smaller particles are called fragments. Nuclear fission is the splitting of a nucleus into smaller particles. Key Terms: Deuterium, Half-Life, Neutron Bombardment, Nuclear Fission, Nuclear Fusion, Nucleus, Radiation, Radioactive Decay, Tritium What is Nuclear Fission  :max_bytes(150000):strip_icc()/artwork-of-nuclear-fusion-reaction-160936093-57f174485f9b586c35490999.jpg)
What is the Difference Between Nuclear Fission and Fusion The main difference between nuclear fission and fusion is that nuclear fission is the division of an atom into smaller particles whereas nuclear fusion is the combination of smaller atoms to form a large atom. Although nuclear fission reactions are not that much found in the environment, nuclear fusion is found in stars such as the sun. Nuclear fusion releases a higher energy than that of nuclear fission. In both reactions, the atoms are altered, and the end products would be completely different from the initial reactants. These reactions release a very high amount of energy. Nuclear fusion and nuclear fission are chemical reactions that take place in the nucleus of an atom. Main Difference – Nuclear Fission vs Fusion
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
